FAQs on Genea Biomedx


Our benchtop incubator — Geri® — is designed to provide individualized and stable culture conditions helping to achieve an optimal environment to improve embryo viability and quality.
- Undisturbed incubation: Geri® has six, individual, single-patient chambers, each independently controlled.
- Fail-safe mechanisms: Safety features, redundancies and alarm functions ensure that stable conditions are maintained.
- Monitor incubation conditions in real time: Individual sensors in each chamber enable monitoring of critical parameters within the incubator
- Improved lab efficiency: Geri® is designed as an easy-to-use, compact, benchtop incubator with a minimal footprint for easy integration into your lab.
- Integrated embryo monitoring: A dedicated high-resolution camera in each chamber provides time-lapse views of each embryo’s development.
- Modular software offer: Geri® software is built on a modular approach to allow integration of new functionalities over time.
- Humidified culture: Geri® is the only time-lapse incubator that offers humidified incubation, proven to result in optimal clinical outcomes
Created after several years of intensive development, Gavi® is the first technology to automate the critical equilibration step, helping to standardize the complex vitrification process. Gavi® approaches vitrification in an entirely new way, helping to achieve consistent high-quality results every time.
- Consistent and reproducible process: A process that happens the same way every time helping to reduce outcome variation across users.
- Unique Gavi® Pod: Innovatively designed to hold oocytes, zygotes, cleavage and blastocyst stage embryos throughout the full vitrification process and storage.
- Precise equilibration process: Gavi® automates and controls critical parameters of temperature, time of exposure, volume and flow rate during the equilibration.
- High-quality results: Blastocyst survival rates exceed competency values defined in the ESHRE/ALPHA Vienna consensus publication on lab performance indicators.
- A controlled, closed-system environment that prevents direct contact between the embryo and liquid nitrogen (LN2).
- Short learning curve: Gavi® requires low technical skills and short time to achieve competency.
Please contact your local Embryologist from Genea Biomedx or one of our authorised distributors to organise a demonstration in your laboratory or in our facility in Madrid.
For more information, We do have demo videos for each product, click on the following links for more information:

Geri®
Award-winning benchtop incubator with continuous embryo monitoring system, with the option of culturing in humid or dry environment.
Learn More
Private: Gavi®
Learn More
Gidget®
A comprehensive witnessing and ART workflow management system designed to aid quality control and improve lab efficiencies
Learn More
Gems®
A complete embryo medium suite, including the third generation of the world’s first sequential media
Learn MoreTo request a quote for an instrument or media, please contact your local Genea Biomedx representative or one of our authorised distributors, or send an email to [email protected]
To talk to one of our sales team please contact your local Genea Biomedx representative, visit our distributor page, or email us on [email protected].
Consumables are used with Geri® and Gavi®. Gavi also comes with supplied accessories.
The Gavi® Family
Gavi® comes with a range of supplied accessories including:

Gavi® Operating Tray
Holds the Gavi® Medium Cartidge, Cassete and Tip & Seal Catridge.

Gavi® LN2 Bucket
Holds liquid nitrogen for the final stage of the vitrification process.

Gavi® Tweezers
For a secure grasp of the Gavi® Cassette when dunked into Gavi® LN2 Bucket.
Consumables used with Gavi®

Gavi® Tip & Seal Cartridge
Holds the disposable pipette tip and Pod lid seal.

Gavi® Medium Cartridge
Contains the Gems® vitrification solutions.

Gavi® Cassette
Holds up to four Pods at once, allowing you to vitrify four blastocysts or 8 oocyte/cleavage stage embryos simultaneously

Gavi® Pod
The Gavi Pod is a container with the capacity to hold two oocytes or zygote/cleavage stage embryos or one blastocyst stage embryo during the vitrification, storage and warming processes.
Accessories used with Gavi®

Gavi® Working station
Holds the Gavi® cassette within LN2 to facilitate the removal of single Pods for thawing.

Gavi® Storage Dividers
For use with cryogenic storage canisters, to provide orderly organization of Cassette and Pods by patient. Available in round or rectangular form.
Geri® consumables and accessories:
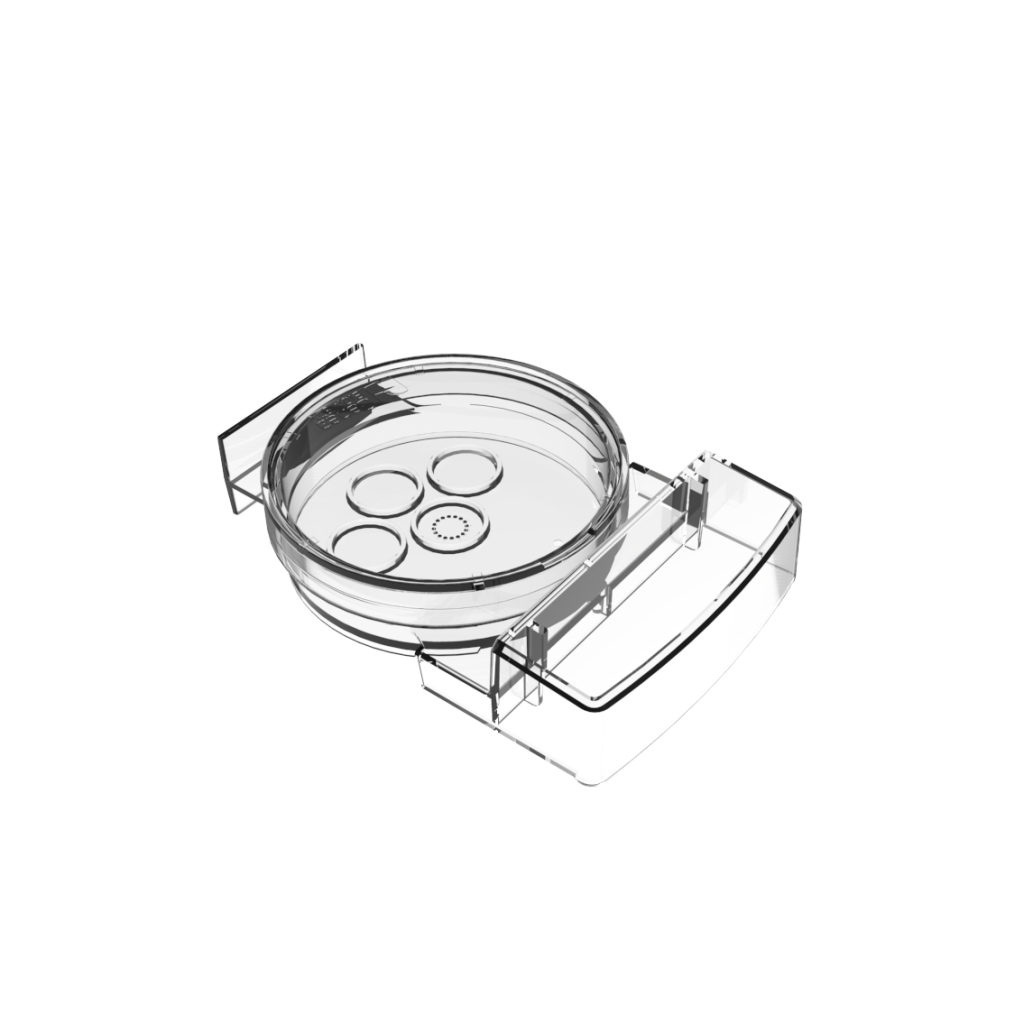
Geri® dish
The Geri dish is a polystyrene, non-pyrogenic device used as an embryo culture dish with the Geri Embryo Incubator for the storage of oocytes or embryos during incubation.

Geri® Water Bottle
The Geri® water bottle is a single use consumable to be used in a single Geri chamber. Correct use of the Geri water bottle will provide a humidified environment inside a Geri chamber.
Please see our distributor page, At this stage we are distributing to the countries marked in gold.
Purchase agreements are set up between clinics and Genea Biomedx, after which orders can be placed by sending an email to our orders teams. Invoices are provided following delivery of all orders.
If you have any issues with the product our engineers will be able to assist. Please contact your sales representative or email [email protected].
We offer a range of written and video training resources to help you quickly reach optimal performance when using Geri® and Gavi® instruments. We have engineers and technical support staff to provide in-person or virtual demonstrations and training
Our time-lapse incubator, Geri®, is a type of incubator designed to provide time-lapse images of embryos as they develop in real-time. Taking pictures every 5 minutes, the purpose of Geri® is to help specialists select the embryos that are most likely to develop into a baby, while keeping the embryos as safe as possible. Geri® is the only humidified time-lapse incubator on the market, further contributing to optimal clinical outcomes, and offers completely undisturbed, individualised patient culture.
IVF involves several steps
- Ovarian stimulation
- Egg retrieval
- Sperm retrieval
- Fertilization
- Embryo transfer.
Gavi® pods are securely stored on cassettes in specially designed single or double level liquid nitrogen Gavi® Storage Dividers. The dividers come in square or round configurations to fit easily into your existing storage vessels.
After the frozen samples have been thawed using the recommended/clinical process, the samples can then be placed into the Geri® for further culture.
This depends on the clinical protocol of the individual clinic. It also depends on the culture media used and most importantly the law/ethics which need to be adhered to for that particular country. In most cases the embryos can remain in the Geri® until that time that the embryo is suitable for transfer or freezing, which is day 5 or 6.
